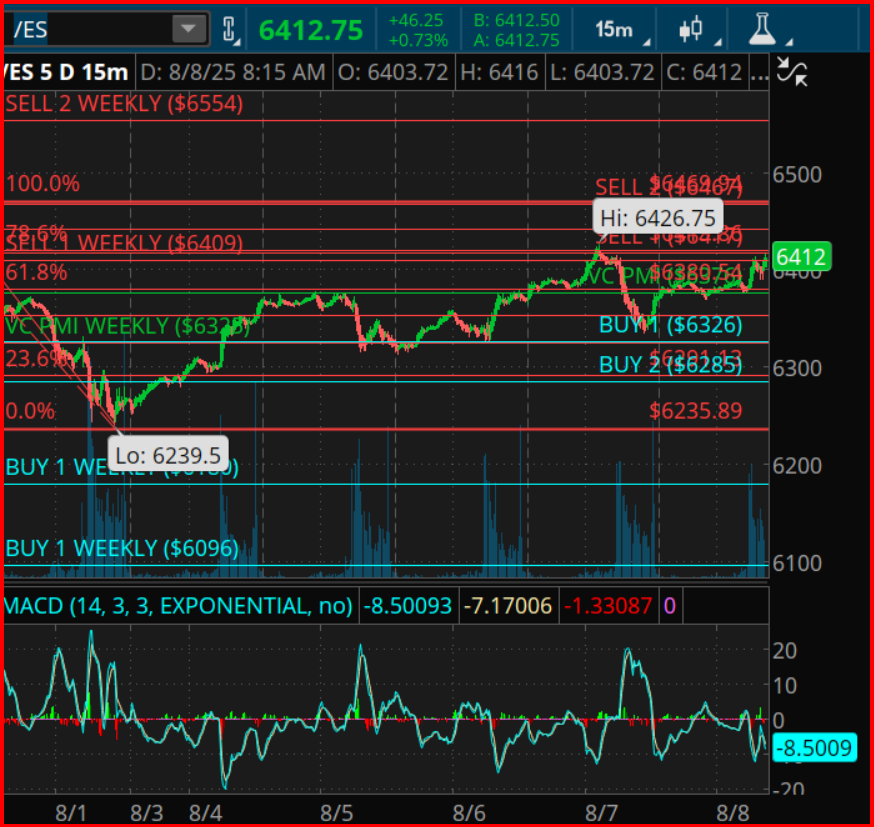
In an intricate dance of market dynamics, the E-mini S&P 500 futures have encountered a pivotal phase where the confluence of price, time, and cyclical analysis presents a profound decision-making juncture for investors. This critical crossroads is dictated by a series of analytical tools that, when combined, offer a glimpse into the potential trajectories of market movements.
To unravel the complexities of this situation, it’s essential to first understand the bullish and bearish paths that lay ahead. The bullish trajectory, for instance, is underscored by a clear continuation pattern. If the market manages to conclude above a specific threshold, it could unlock further ascending strides towards subsequent resistance levels delineated by Fibonacci retracement calculations and harmonic analysis.
On the contrary, the narrative also outlines a well-defined bearish reversal route. Should the market falter, falling below a pivotal support level, it would signify the unravelling of the bullish structure. This retreat could initiate a descent towards a retest of previous lows, setting the stage for a potential cyclical downturn as outlined by historical market rhythms.
Adding to the gravity of the current juncture is the 360-day master cycle, a temporal framework that accentuates the significance of the timing window between August 9th and August 14th. This specific period does not represent merely another fleeting moment in market analysis. Rather, it is underscored by historical tendencies that often herald a turning point in market trends, propelling a shift that could define subsequent trading quarters. Historically, this phase often precedes a directional shift, marking a cyclical crest that could lead to a seasonal correction as past patterns have shown.
To contextualize, the E-mini S&P 500 futures have been on an upward trajectory since the commencement of August, achieving substantial gains. It is during this ascent that traders have witnessed breaks through critical resistance levels, underpinned by analytical tools such as the Fibonacci retracements and harmonic squares. The market’s ability to sustain above these levels could signal further bullish momentum, potentially leading to new heights.
However, it is critical to approach this juncture with heightened scrutiny. As the market moves towards the specified dates in August, each increment in price and time must be meticulously analyzed through the lens of the aging cyclical rhythm. The implications of crossing above or below certain thresholds are profound, delineating paths of continuation or reversal that are deeply rooted in historical market behavior.
In simple terms, the E-mini S&P 500 futures stand at a juncture teeming with potential for both a bullish breakout and a cyclical downturn. The decisions made in this compressed timeframe could forge the direction for the remainder of the quarter, making this an opportune moment for precision-driven trading and disciplined timing.
It is against this backdrop that traders and investors are reminded of the inherent risks associated with trading derivatives, financial instruments, and precious metals. The volatile nature of these markets demands a strategic approach, underscored by an understanding that past performance is not always indicative of future results.
As we navigate through this critical stretch, it becomes imperative to remain engaged, harnessing technical acumen in tandem with an understanding of market cycles to make informed decisions. This convergence of analytical tools serves not just as a technical exercise, but as a strategic fulcrum, potentially guiding the trajectory of the E-mini S&P 500 futures in the weeks to come.